
… The beta pleated sheet is polypeptide chains running along side each other. This allows all amino acids in the chain to form hydrogen bonds with each other. The alpha helix is formed when the polypeptide chains twist into a spiral. What do alpha helices and beta sheets do? … Parallel sheets are less twisted than antiparallel and are always buried. However, most beta-sheets found in globular protein X-ray structures are twisted. The Pauling-Corey model of the beta-sheet is planar. Sheets tend to be either all parallel or all antiparallel, but mixed sheets do occur. What is the MOST important factor that drives the formation of secondary structure of proteins? Are beta sheets always antiparallel? Which is the more stable Beta-sheet: parallel or antiparallel? Antiparallel due to more linear H-bonds and a high concentration of Ala and Gly. Read More: What ocean basin is Agulhas in? Which is the more stable beta-sheet parallel or antiparallel quizlet? Antiparallel hairpins are disfavored compared to beta-sheets formed by sequentially separated strands. … Parallel beta-sheets and their subunits are energetically less stable and indeed found to occur more rarely in proteins. Are beta pleated sheets stable?īeside alpha-helices, beta-sheets are the most common secondary structure elements of proteins. This is the main difference between Alpha Helix and Beta Pleated Sheet. groups in the backbone of the adjacent strands. 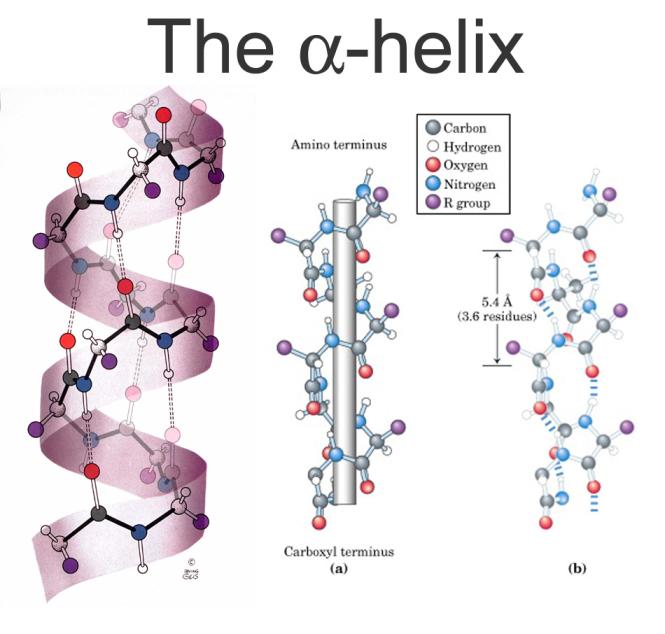 
In contrast to the alpha helix, hydrogen bonds in beta sheets form in between N-H groups in the backbone of one strand and C=O. What are the differences between α helix and β pleated sheet structure of protein? … Explanation: A coiled peptide chain held in place by hydrogen bonding between peptide bonds in the same chain is α helix. Which of the following does not affect the stability of an α-helix? Explanation: The occurrence of Proline and Glycine residues affect the stability of an α-helix. Hence, option b is correct.Which of the following does not affect the stability of an α helix?ġ. In a β-pleated sheet, polypeptide units lying adjacent to each other form hydrogen bonds giving it a folded or pleated appearance. In an α-helix, the carbonyl group of one amino acid forms a hydrogen bond with an amino acid that is four down the polypeptide chain. Both these structures are held in shape by hydrogen bonding between the carbonyl group of one amino acid and amino groups of another amino acid. The two most well-known secondary structures are α-helix and the β-pleated sheet. The secondary structure of a protein refers to the folding of the polypeptide backbone. Each polypeptide chain is referred to as a subunit that arranges itself to form the multi-subunit protein. Hence, option c is incorrect.Ī quaternary structure is formed when the three-dimensional structures of multiple polypeptide chains interact with one another. Hydrogen bonding, ionic bonding, and disulfide bridges also contribute to the folding and stabilization of the protein structure. The hydrophilic R groups are exposed to the aqueous environment whereas the hydrophobic R groups position themselves at the center of the protein. Hydrophobic interactions play a major role in the folding of the protein. Several forces hold the tertiary structure of the protein. The tertiary structure is formed when the polypeptide chain folds to give a protein its unique three-dimensional shape for optimum function. The primary structure of a protein is the unique linear order of amino acids linked together by peptide bonds to form the polypeptide chain.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
